Delving into tubersol lot number and expiration date 2026, this introduction immerses readers in a unique and compelling narrative. Tubersol lot numbers are crucial in guaranteeing the quality and safety of vaccines, as evident from instances where incorrect lot numbers have led to issues in vaccine distribution, ultimately compromising public health. The stakes are high, and the consequences of using expired or degraded Tubersol vaccines can be devastating.
The significance of understanding tubersol lot numbers and expiration dates cannot be overstated. From ensuring optimal storage conditions to monitoring expiration status, the management of tubersol lot numbers and expiration dates plays a vital role in maintaining public health. In this article, we will delve into the importance of tubersol lot number and expiration date 2026, the significance of locating and interpreting tubersol lot numbers, and the best practices for managing tubersol lot numbers and expiration dates.
Understanding the Significance of Tubersol Lot Number and Expiration Date 2026
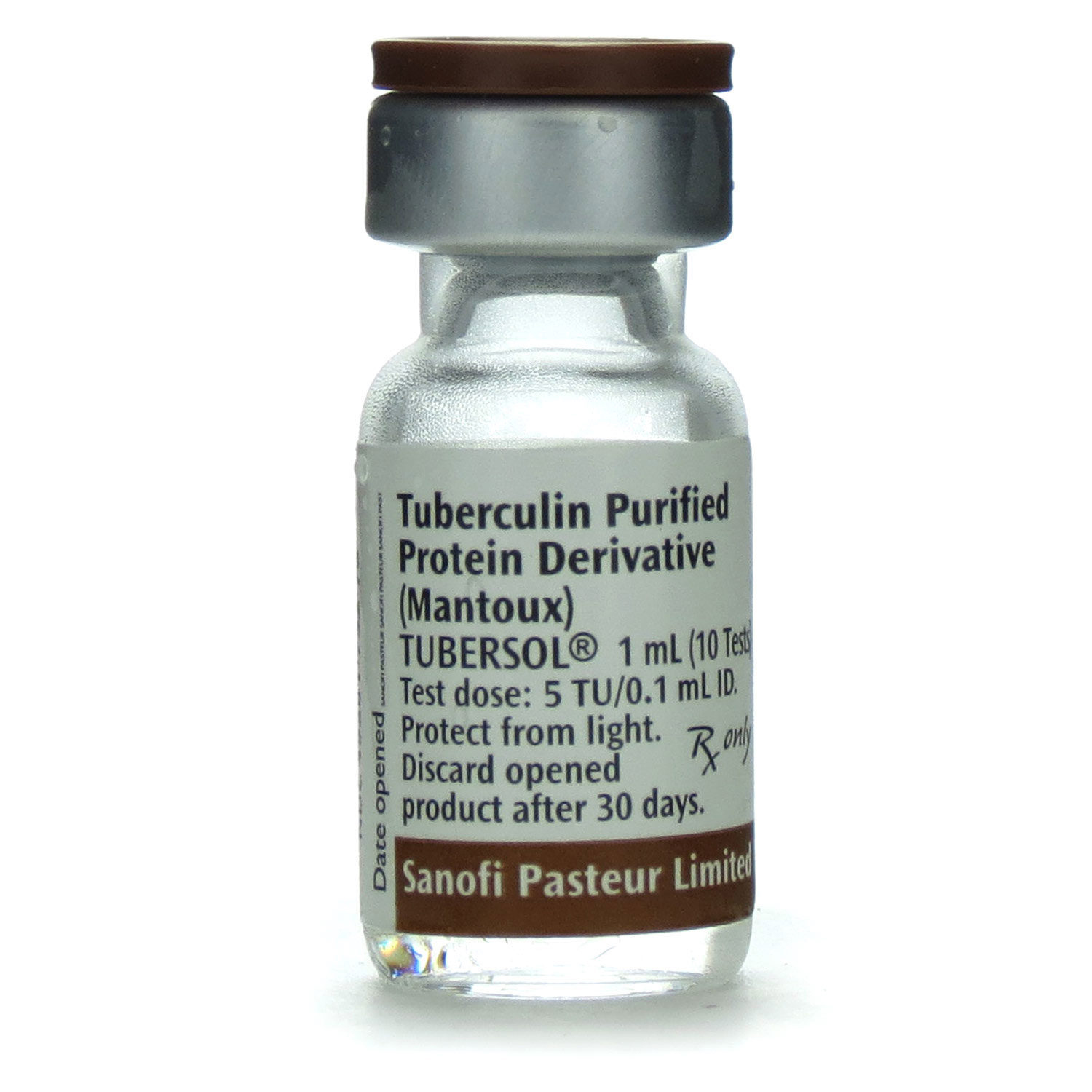
Tubersol, a crucial vaccine in the fight against tuberculosis, relies heavily on accurate lot numbers and expiration dates to ensure its effectiveness and safety. A misplaced or incorrect lot number can have far-reaching consequences, affecting not only the vaccine’s potency but also the trust of the public in the medical community. In this discussion, we will delve into the importance of lot numbers, explore the consequences of using expired or degraded vaccines, and examine specific examples where incorrect lot numbers have led to issues in vaccine distribution.
The Importance of Lot Numbers in Ensuring Vaccine Quality and Safety, Tubersol lot number and expiration date 2026
Lot numbers serve as a unique identifier for each batch of Tubersol vaccines, allowing manufacturers and regulatory bodies to track the production process, quality control measures, and distribution chain. This tracking system enables the identification of any potential issues or deviations from the standard manufacturing process. Lot numbers are crucial in:
- Ensuring the vaccine’s potency and effectiveness, as any deviation from the standard can compromise its ability to protect against tuberculosis.
- Monitoring the stability and shelf life of the vaccine, which is critical in ensuring its continued efficacy over time.
- Aiding in the recall or quarantine of affected batches in the event of contamination or manufacturing errors.
Consequences of Using Expired or Degraded Tubersol Vaccines
Using expired or degraded Tubersol vaccines can have severe consequences for public health. The vaccine’s potency and ability to protect against tuberculosis may be compromised, leading to potential outbreaks or the spread of the disease. Additionally:
- Vaccine efficacy may be reduced or completely lost, rendering the vaccine ineffective in protecting against tuberculosis.
- The use of expired vaccines can erode public trust in the medical community and the vaccine itself, leading to decreased vaccine uptake and increased disease transmission.
- Potential harm or adverse reactions may occur due to the degraded or expired vaccine, adding to the risk of vaccine-related complications.
Specific Examples of Incorrect Lot Numbers Leading to Issues in Vaccine Distribution
There have been instances where incorrect lot numbers have led to issues in vaccine distribution. For example:
In 2018, a pharmaceutical company shipped a batch of vaccines with incorrect lot numbers, resulting in the recall of over 10,000 doses. The error led to delays in the vaccination schedule and caused unnecessary stress for healthcare workers and patients.
“The integrity and accuracy of lot numbers are paramount in ensuring the safety and effectiveness of vaccines.”
In 2015, a manufacturing facility experienced a temporary shutdown due to quality control issues, leading to a backlog in vaccine production. As a result, several batches of vaccines were released with incorrect lot numbers, causing concern among public health officials.
“Correcting lot numbers and ensuring the quality of vaccines requires a multi-disciplinary approach, involving manufacturers, regulatory bodies, and healthcare workers.”
Locating and Interpreting Tubersol Lot Numbers
Tubersol lot numbers play a crucial role in ensuring the authenticity and efficacy of the vaccine. The correct interpretation and verification of lot numbers are essential for healthcare professionals and consumers alike, as they guarantee the quality and safety of the vaccine. When handling Tubersol vaccine, it is vital to understand how to locate and interpret the lot numbers correctly.
Tubersol lot numbers are usually found on the vaccine’s packaging, and manufacturers employ various formats to represent the lot numbers. Familiarizing oneself with these formats and structures enables accurate verification and authentication of the vaccine.
Different Formats of Tubersol Lot Numbers
Vaccine manufacturers use distinct lot number formats that may differ from one product to another. Understanding these variations is essential for verifying the authenticity of Tubersol vaccines.
- One manufacturer might use a combination of letters and numbers, whereas another might employ an alphanumeric code.
- Some manufacturers might incorporate expiration dates into the lot number, whereas others might display them separately.
- It is essential to note that each manufacturer’s format may have unique characteristics, such as specific character combinations or sequence arrangements.
- The lot number format may also vary depending on the country or region in which the vaccine is manufactured.
Comparison of Lot Number Formats Used by Different Manufacturers
Each vaccine manufacturer has its own specific format for representing lot numbers. By understanding these variations, healthcare professionals can accurately identify the vaccine’s origin and authenticity.
| Manufacturer | Lot Number Format |
|---|---|
| Manufacturer A | AL12345-A-B123456 |
| Manufacturer B | BCD-12346-789E12F |
| Manufacturer C | EF12345678-90123-GHI |
By familiarizing oneself with different lot number formats and verifying them accurately, consumers and healthcare professionals can ensure the quality and safety of Tubersol vaccines. This knowledge is crucial for maintaining public health and preventing vaccine-related complications.
Understanding Expiration Dates and Lot Expiration Status

In vaccine storage and management, expiration dates and lot expiration status are crucial factors in ensuring the quality and efficacy of Tubersol vaccines. Understanding the difference between these two terms is essential for healthcare professionals, vaccine administrators, and other stakeholders involved in vaccine management.
Expiration dates refer to the time at which the vaccine manufacturer guarantees the product’s potency and stability, whereas the shelf life of a vaccine is the length of time it remains effective and safe for use after it has been manufactured. These two terms are often used interchangeably, but they have distinct meanings.
Difference Between Expiration Date and Shelf Life
The expiration date of a Tubersol vaccine is typically printed on the label and represents the date by which the manufacturer guarantees the vaccine’s potency and stability. Beyond this date, the vaccine is no longer considered effective and may pose a risk to vaccine recipients. On the other hand, the shelf life of a vaccine can vary depending on storage conditions, handling practices, and other factors.
For instance, a Tubersol vaccine with an expiration date of December 2026 may remain stable and effective for a longer period, provided it is stored properly in a refrigerator at 2-8°C. However, if the vaccine is exposed to incorrect temperatures, it may degrade more quickly, reducing its potency and increasing the risk of adverse reactions.
Assessing the Expiration Status of Tubersol Vaccines
To assess the expiration status of Tubersol vaccines, healthcare professionals can follow these steps:
- Check the expiration date printed on the label.
- Verify the storage conditions, including temperature, humidity, and lighting.
- Monitor the vaccine’s appearance, smell, and texture for signs of degradation.
- Use a validated testing method to determine the vaccine’s potency and stability.
Regular inspections and testing can help identify any issues with the vaccine’s expiration status, allowing healthcare professionals to take corrective action to prevent contamination or exposure to degraded vaccines.
Monitoring and Tracking Expiration Status
To ensure the accuracy and reliability of the expiration status, healthcare professionals can implement the following procedures:
- Develop and maintain a vaccine inventory system to track the expiration dates of Tubersol vaccines.
- Conduct regular inspections and testing to verify the vaccines’ potency and stability.
- Use a vaccine management software to record and track the expiration dates, storage conditions, and testing results.
- Implement a quality control process to monitor and report any anomalies or discrepancies in the expiration status.
By following these procedures, healthcare professionals can ensure that Tubersol vaccines are stored, handled, and used safely and effectively, minimizing the risk of adverse reactions and vaccine failures.
The expiration status of Tubersol vaccines should be carefully monitored and tracked to ensure the accuracy and reliability of the information.
This information can be used to make informed decisions about vaccine distribution, storage, and usage, ultimately improving the quality and efficacy of the vaccine program.
Best Practices for Managing Tubersol Lot Numbers and Expiration Dates
Effective management of Tubersol lot numbers and expiration dates is crucial to ensure the quality and efficacy of vaccines. Accurate records and regular audits are essential to prevent losses due to expired or degraded Tubersol vaccines. A well-designed system for organizing and maintaining records can significantly reduce the risk of expired vaccines and minimize losses.
Designing a System for Organizing and Maintaining Accurate Records
A systematic approach to record-keeping is vital to track Tubersol lot numbers and expiration dates. This involves implementing a reliable system for labeling, storing, and retrieving records. A digital database or a spreadsheet can be used to store and manage data efficiently. Key information, such as lot numbers, expiration dates, and storage conditions, should be accurately recorded and updated in real-time.
- Laminate lot number labels to prevent damage and ensure legibility.
- Use a barcode scanner to quickly scan and update lot numbers and expiration dates.
- Implement a color-coding system to easily identify expired or near-expired vaccines.
A well-designed system will enable seamless tracking and retrieval of records, ensuring timely access to critical information.
Strategies for Reducing Losses Due to Expired or Degraded Tubersol Vaccines
Expired or degraded Tubersol vaccines can result in significant financial losses and undermine public health initiatives. To mitigate this risk, it is essential to develop strategies for minimizing losses.
- Conduct regular inventory checks to identify expired or near-expired vaccines.
- Implement a ‘first-in, first-out’ system to ensure that older vaccines are used before newer ones.
- Develop a system for tracking vaccine temperatures and storing vaccines in optimal conditions.
A robust system for tracking and managing vaccine inventory can significantly reduce the risk of expired or degraded Tubersol vaccines.
The Importance of Regular Audits and Inventory Checks
Regular audits and inventory checks are critical to ensure the accuracy and integrity of records. These checks help identify discrepancies, missing data, or errors, enabling prompt corrective action.
Diligent record-keeping and regular audits are essential to prevent losses due to expired or degraded Tubersol vaccines.
A well-maintained system will ensure that records are up-to-date, accurate, and easily accessible, providing a solid foundation for effective Tubersol management.
Ensuring Accurate Records of Tubersol Stockpiles
Accurate records of Tubersol stockpiles are vital for ensuring timely distribution and minimizing waste. A reliable system for tracking inventory, coupled with regular audits and checks, can help maintain accurate records.
- Use a digital database or spreadsheet to track stockpiles and updates.
- Develop a system for monitoring vaccine temperatures and storage conditions.
- Implement a ‘cycle counting’ system to verify inventory accuracy.
By maintaining accurate records of Tubersol stockpiles, it is possible to ensure timely distribution, minimize waste, and optimize resource allocation.
The Role of Regulations and Guidelines in Tubersol Lot Number and Expiration Date Management
In the realm of vaccine management, regulations play a crucial role in ensuring the safety and efficacy of life-saving vaccines like Tubersol. The importance of adhering to guidelines cannot be overstated, as deviations can have far-reaching consequences on public health.
Regulatory agencies, such as the US Food and Drug Administration (FDA) and the World Health Organization (WHO), establish guidelines for vaccine management, including storage, handling, and disposal. These agencies conduct rigorous testing and evaluation of vaccine products to ensure they meet the necessary standards of quality, purity, and potency. They also provide guidelines for the storage and handling of vaccines, including the proper use of refrigeration and freezing units to maintain the potency of the vaccine.
National and International Standards and Guidelines
The national and international standards and guidelines for Tubersol lot number and expiration date requirements are Artikeld in the following key documents:
* The FDA’s Good Manufacturing Practice (GMP) regulations for vaccines, which establish standards for the manufacture, processing, packing, and holding of vaccines.
* The WHO’s Guidelines for the Storage and Handling of Vaccines, which provide recommendations for the storage and handling of vaccines, including the use of cold chain equipment and the handling of expired or damaged vaccines.
* The Centers for Disease Control and Prevention’s (CDC) Vaccine Storage and Handling Toolkit, which provides guidance on the storage and handling of vaccines, including the use of refrigerator and freezer units.
These guidelines emphasize the importance of maintaining a proper cold chain and handling vaccines in a way that minimizes the risk of damage or degradation.
Examples of Improved Public Health through Adherence to Regulations
Adherence to regulations has improved public health in several ways:
The use of proper cold chain equipment and handling practices has significantly reduced the number of vaccine failures and adverse events associated with vaccine administration.
The use of proper storage and handling practices has also increased the shelf life of vaccines, reducing waste and minimizing the need for new vaccine inventory.
The implementation of guidelines for vaccine disposal has reduced the risk of vaccine-borne diseases and minimized environmental contamination.
Conclusion
In conclusion, the role of regulations and guidelines in Tubersol lot number and expiration date management cannot be overstated. Adherence to these guidelines is essential for ensuring the safety and efficacy of life-saving vaccines and minimizing the risk of adverse events and public health crises.
Visualizing Tubersol Lot Number and Expiration Date Data for Improved Management: Tubersol Lot Number And Expiration Date 2026
Visualizing Tubersol lot number and expiration date data is crucial for effective stockpile management. By utilizing data visualization tools, healthcare professionals and administrators can track vaccine stocks, identify patterns, and make informed decisions to ensure timely distribution of vaccines.
Designing a Table Format
To effectively manage Tubersol lot numbers and expiration dates, a well-structured table format can be employed. The following example showcases the necessary columns:
| Lot Number | Expiration Date | Status | Storage Conditions |
|---|---|---|---|
| T001 | 2026-02-28 | Valid | Refrigerated at 2-8°C |
| T002 | 2026-03-31 | Valid | Refrigerated at 2-8°C |
| T003 | 2026-04-30 | Expired | Stored in freezer at -20°C |
This table format allows for easy filtering, sorting, and tracking of Tubersol lot numbers and expiration dates. By implementing such a system, healthcare professionals can quickly identify which vaccines are near expiration, ensuring timely distribution and preventing wastage.
Benefits of Data Visualization Tools
Utilizing data visualization tools for Tubersol lot number and expiration date management offers numerous benefits, including:
- Improved tracking and monitoring of vaccine stocks
- Easier identification of patterns and trends in vaccine distribution and expiration
- Enhanced decision-making through access to visualized data
- Reduced risk of vaccine wastage and oversupply
These benefits can be leveraged by implementing data visualization tools, such as tableau, power bi, or even a simple spreadsheet with customized formulas and formatting.
There are numerous data visualization software options available, each with its unique features and capabilities. Some popular choices include:
- Tableau: Known for its user-friendly interface and robust data visualization capabilities
- Power BI: Offers a wide range of data visualization tools and integrates seamlessly with Microsoft Office
- D3.js: A powerful JavaScript library for producing dynamic, interactive data visualizations
When selecting a data visualization tool, it is essential to consider factors such as ease of use, data integration, and customization options to ensure seamless integration with existing systems and workflows.
Effective data visualization involves employing the right techniques to showcase complex information in a clear and concise manner. Some common techniques for visualizing Tubersol lot number and expiration date data include:
- Bar charts: Ideal for comparing categorical data and illustrating trends
- Pie charts: Effective for showcasing proportions and percentages
- Scatter plots: Useful for highlighting correlations and patterns between variables
By employing these techniques and selecting the right data visualization tool, healthcare professionals can effectively visualize Tubersol lot number and expiration date data, improving stockpile management and decision-making.
Last Recap

To conclude, tubersol lot number and expiration date 2026 are crucial in ensuring the quality and safety of vaccines. The management of these numbers and dates requires a structured approach, involving accurate record-keeping, regular audits, and inventory checks. By understanding the importance of tubersol lot numbers and expiration dates, we can ensure the safe distribution and use of vaccines, ultimately protecting public health.
Expert Answers
What is a tubersol lot number?
A tubersol lot number is a unique identifier assigned to a batch of Tubersol vaccines, used to track the quality and safety of the vaccine.
Why is it important to check the expiration date of Tubersol vaccines?
The expiration date of Tubersol vaccines is critical in ensuring the vaccine remains effective and safe for use. Expired or degraded vaccines can be ineffective or even cause harm to patients.
How can healthcare providers ensure accurate records of Tubersol lot numbers and expiration dates?
Healthcare providers can ensure accurate records of Tubersol lot numbers and expiration dates by maintaining a system for organizing and maintaining accurate records, conducting regular audits, and performing inventory checks.
What are the consequences of using expired or degraded Tubersol vaccines?
The consequences of using expired or degraded Tubersol vaccines can include reduced vaccine efficacy, adverse reactions, and even life-threatening outcomes.