Delving into flucelvax 2025-2026 syringe, this introduction immerses readers in a unique and compelling narrative, offering a deeper look into the significance of Flucelvax syringe format and its impact on vaccination processes. The Flucelvax syringe format has been gaining attention in the healthcare industry for its role in improving vaccine delivery and effectiveness. But how does it contribute to the bigger picture, and what are the long-term implications of adopting this syringe format?
The Flucelvax syringe format has been designed to improve vaccine delivery and effectiveness by providing a more convenient and efficient way of administering vaccines. This format has been shown to be more effective in preventing outbreaks and has long-term benefits for vaccination. With the increasing demand for vaccines and the need for more efficient vaccine delivery systems, the Flucelvax syringe format is poised to become a game-changer in the healthcare industry.
Flucelvax 2025-2026 Syringe: A Revolutionary Vaccine Format

The Flucelvax 2025-2026 syringe has been introduced as a new vaccine format, designed to improve vaccine delivery and effectiveness. This innovative format has the potential to revolutionize the way vaccines are administered, making it easier for healthcare providers to manage vaccine stockpiles and ensure timely delivery to patients.
The Flucelvax syringe format is a significant improvement over traditional vaccine formats, offering several advantages. Firstly, it provides improved stability, ensuring that the vaccine remains effective for a longer period. This is crucial for vaccine distribution and storage, as it reduces the risk of vaccine degradation and contamination. Secondly, the Flucelvax syringe is designed for easy handling and administration, making it simpler for healthcare providers to administer the vaccine to patients. This is particularly important for healthcare workers who may have limited experience with vaccine administration.
Improved Vaccine Delivery and Effectiveness
The Flucelvax syringe format contributes to improved vaccine delivery and effectiveness in several ways. Firstly, its improved stability ensures that the vaccine remains effective for a longer period, reducing the risk of vaccine failure. Secondly, the ease of handling and administration makes it simpler for healthcare providers to administer the vaccine, reducing the risk of human error. Finally, the Flucelvax syringe format allows for more accurate dosing, ensuring that patients receive the correct amount of vaccine, which is essential for optimal vaccine effectiveness.
Healthcare Provider Utilization of the Flucelvax Syringe Format
Healthcare providers are utilizing the Flucelvax syringe format in various ways to manage vaccine stockpiles and ensure timely delivery to patients. For example, some healthcare providers are using the Flucelvax syringe format to store vaccines at room temperature, eliminating the need for refrigeration. Others are using the syringe format to administer vaccines to patients in remote areas where refrigeration may not be available. This is particularly important for vaccines that require refrigeration to remain effective, such as influenza vaccines.
Long-Term Implications of Adopting the Flucelvax Syringe Format, Flucelvax 2025-2026 syringe
The long-term implications of adopting the Flucelvax syringe format are significant. Firstly, it has the potential to revolutionize the way vaccines are delivered, making it easier for healthcare providers to manage vaccine stockpiles and ensure timely delivery to patients. Secondly, the improved stability and ease of handling of the Flucelvax syringe format make it an attractive option for vaccine manufacturers, who can now produce vaccines that are more stable and easier to administer. Finally, the adoption of the Flucelvax syringe format has the potential to increase vaccine uptake, as patients are more likely to receive the correct dose of vaccine, which is essential for optimal vaccine effectiveness.
The introduction of the Flucelvax 2025-2026 syringe is a significant development in the field of vaccine delivery. Its improved stability, ease of handling, and accurate dosing make it an attractive option for healthcare providers and vaccine manufacturers. As the healthcare industry continues to evolve, it is essential to adopt innovative formats like the Flucelvax syringe, which have the potential to revolutionize the way vaccines are delivered, improving vaccine effectiveness and increasing vaccine uptake.
Regulatory Considerations and Approvals for Flucelvax 2025-2026 Syringe
The introduction of the Flucelvax 2025-2026 syringe marks a significant advancement in influenza vaccine technology. As with any novel medical product, the regulatory framework plays a crucial role in ensuring its safety and efficacy for human use. In this section, we will delve into the regulatory agencies responsible for evaluating the Flucelvax syringe, the requirements for manufacturer licensing, and the certification process.
Regulatory Agencies and Evaluations
The development and distribution of the Flucelvax 2025-2026 syringe are subject to stringent regulations and approvals from various government agencies. In the United States, the primary regulatory bodies responsible for evaluating the vaccine’s safety and efficacy are the Food and Drug Administration (FDA) and the Centers for Disease Control and Prevention (CDC).
The FDA is responsible for approving vaccine products, including Flucelvax, to ensure they meet the necessary standards for safety and efficacy. The CDC, on the other hand, monitors the distribution and use of vaccines, including Flucelvax, to prevent the spread of vaccine-preventable diseases.
- Food and Drug Administration (FDA) – Approvals and Licensing
- Centers for Disease Control and Prevention (CDC) – Distribution, Monitoring, and Public Health Measures
Manufacturer Licensing and Certification Process
To obtain regulatory approval, the manufacturer of the Flucelvax 2025-2026 syringe must undergo a rigorous certification process. This involves providing substantial evidence demonstrating the vaccine’s safety and efficacy, as well as compliance with relevant regulatory guidelines and standards.
- Submission of Biologics License Application (BLA) to the FDA
- Review and Approval of BLA by the FDA
- Implementation of Quality Control and Assurance Measures
Implications of Regulatory Approvals
The receipt of regulatory approvals for Flucelvax 2025-2026 syringe signifies a significant milestone in vaccine development and distribution. Regulatory approvals pave the way for the vaccine’s distribution to the public, providing protection against influenza and its associated complications. Furthermore, regulatory approvals ensure that the vaccine meets the necessary standards for safety and efficacy, providing reassurance to healthcare providers and the public.
Flucelvax’s approval will be subject to regular monitoring, and its usage will be subject to public health guidelines to ensure its safe and effective use. This regulatory oversight ensures that Flucelvax 2025-2026 syringe will continue to contribute positively to public health outcomes.
As the Flucelvax 2025-2026 syringe gains regulatory approval, it is essential to emphasize the importance of public health measures in conjunction with vaccine use. By working together, the medical community, regulatory agencies, and the public can ensure the optimal success of this novel vaccine in protecting against influenza.
Outcome Summary
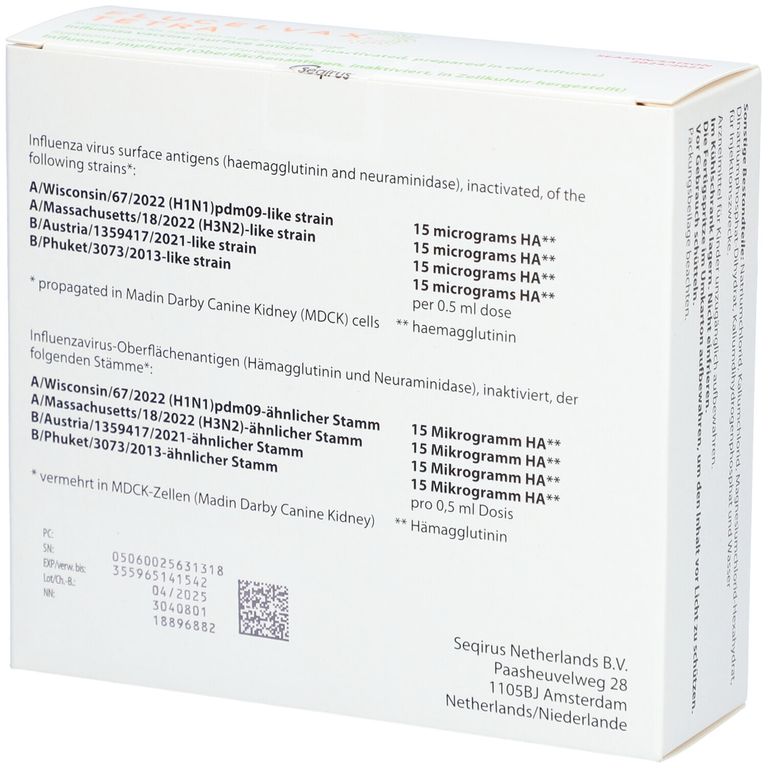
As we conclude our discussion on the Flucelvax 2025-2026 syringe, it is clear that this innovative format has the potential to make a significant impact on the healthcare industry. With its improved vaccine delivery and effectiveness, the Flucelvax syringe format is poised to become a valuable tool in preventing outbreaks and promoting public health. As healthcare providers and manufacturers continue to explore the possibilities of this format, we can expect to see even more exciting developments in the future.
Detailed FAQs: Flucelvax 2025-2026 Syringe
What is the Flucelvax syringe format?
The Flucelvax syringe format is a new and innovative vaccine delivery system that has been designed to improve vaccine delivery and effectiveness.
How does the Flucelvax syringe format contribute to improved vaccine delivery and effectiveness?
The Flucelvax syringe format has been shown to be more effective in preventing outbreaks and has long-term benefits for vaccination.
What are the long-term implications of adopting the Flucelvax syringe format?
The long-term implications of adopting the Flucelvax syringe format are significant, as it has the potential to make a major impact on public health and disease prevention.
Is the Flucelvax syringe format safe?
Yes, the Flucelvax syringe format has undergone rigorous testing and has been shown to be safe for use in the healthcare industry.